 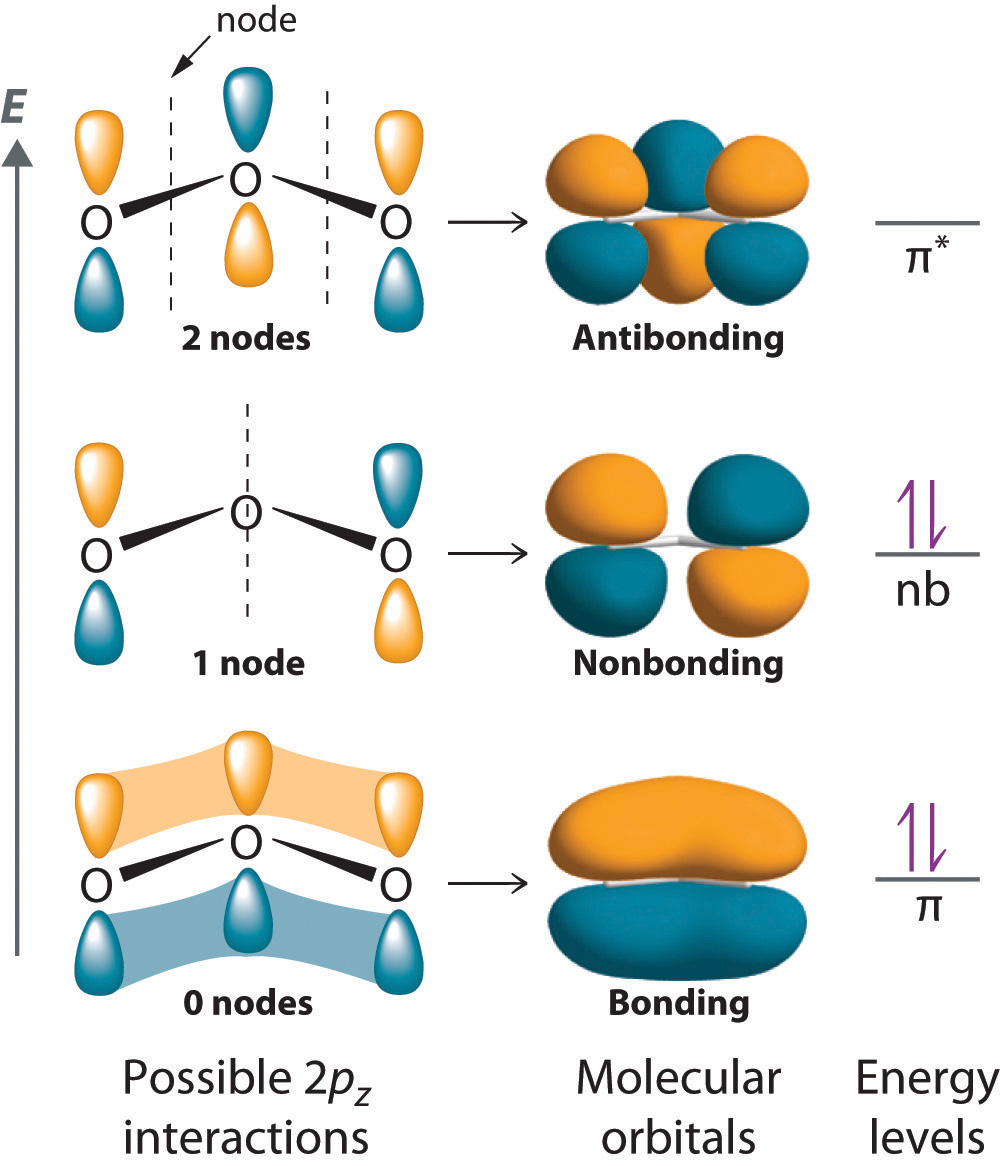
Furthermore, electrons may occupy antibonding orbitals if the molecule is in the excited state. 
Bonding molecular orbitals contain electrons in the ground state while antibonding molecular orbitals contain no electrons in the ground state. Moreover, there are two types of molecular orbitals: bonding molecular orbitals and antibonding molecular orbitals. Similar to atomic orbitals, molecular orbitals maximally contain 2 electrons, which have opposite spins.įigure 01: Molecular Orbitals in a Molecule Furthermore, the molecular orbital surrounds the two nuclei of the atoms, and electrons can move around both nuclei. The number of newly formed molecular orbitals is equal to the number of combined atomic orbitals. When two atoms move closer together to form a molecule, atomic orbitals overlap and combine to become molecular orbitals. Side by Side Comparison – Molecular Orbital vs Atomic Orbital in Tabular FormĪtoms join together to form molecules. Wave function (Ψ) corresponds to different states for the electron. With this, Schrodinger developed equations to find the wave nature of an electron and came up with the wave equation and wave function. When quantum mechanics came into the picture with their findings, it was discovered that an electron has both particle and wave properties. The bonding in molecules was understood in a new way with the new theories presented by Schrodinger, Heisenberg and Paul Dirac. 
The key difference between molecular orbital and atomic orbital is that atomic orbitals describe the locations where the probability of finding the electrons is high in an atom whereas molecular orbitals describe the probable locations of electrons in a molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
